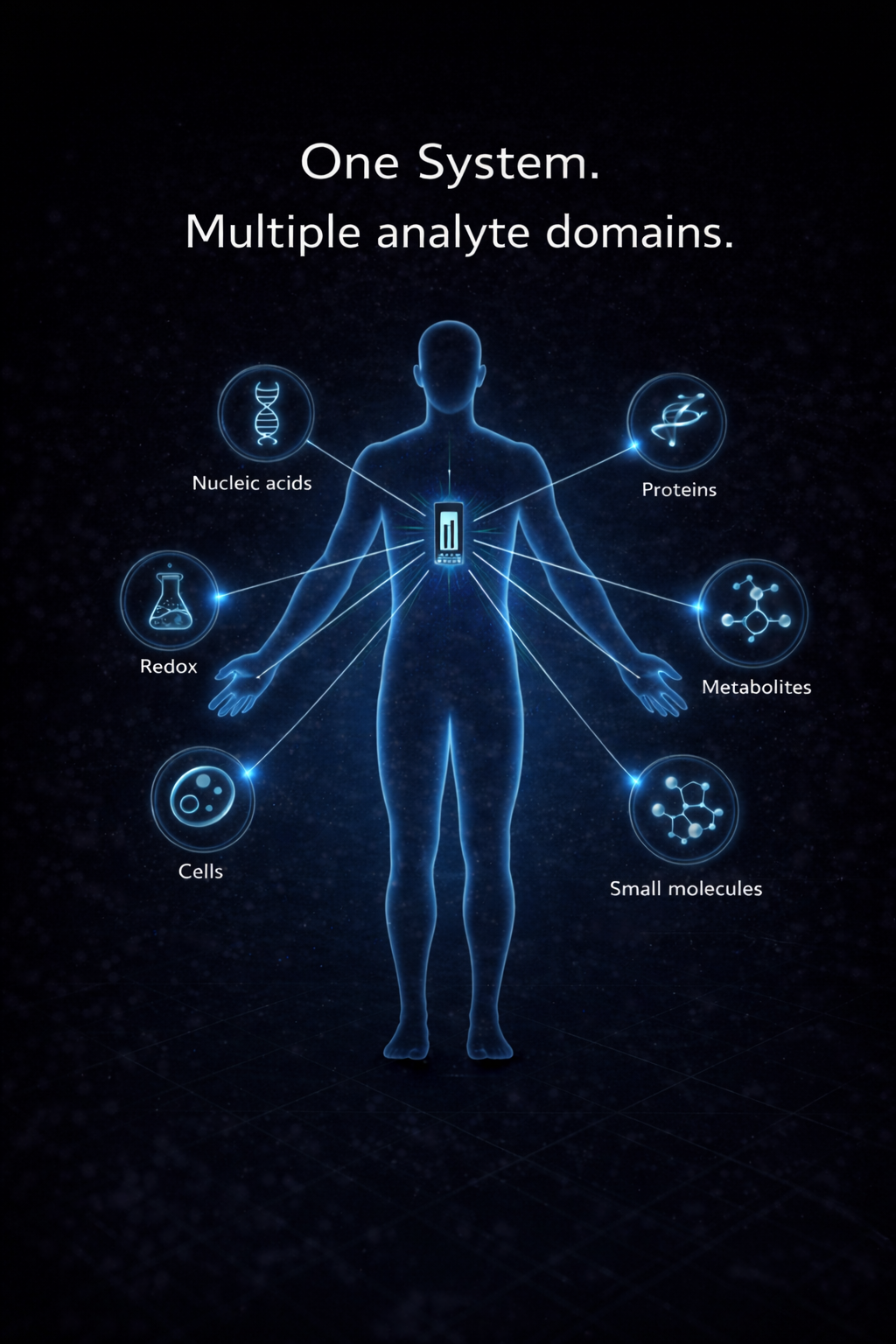
Build assays that existing platforms cannot support
Advanced assays fail when measurement systems cannot capture sparse, dynamic, multi-dimensional biology within practical cost and deployment constraints.
Guanine replaces instrument-by-instrument development with a shared measurement architecture—allowing OEMs to develop advanced assays without building new systems for each biomarker or workflow.
Simple assays are solved. Advanced assays are not.
Conventional systems were built for abundant, isolated, static measurements. Advanced assays are sparse, low-level, multi-analyte, and often time-dependent.
That is why each new advanced assay often requires a new instrument, a new workflow, and a new validation path. The bottleneck is not biology. It is the measurement architecture.
Low-abundance signals
Signal is too weak for practical deployment on many existing systems.
Rare targets in large samples
Targets are too dilute to recover efficiently with small-volume workflows.
Time-dependent biology
Static endpoint systems cannot capture dynamic treatment response or cellular behavior.

Different applications. The same measurement limitation.
What appear to be separate markets are different expressions of the same system failure: existing platforms do not scale with biological complexity.
Low-Abundance Proteins
Host-response proteins and subtle biomarker panels at low concentration.
Rare Cells & Pathogens
Dilute targets in large samples, including bloodstream infections and rare cell capture.
Genetic Panels
Multiplex nucleic acid and resistance-marker measurement without system expansion.
Multi-Analyte Panels
Host, pathogen, protein, nucleic acid, and other analytes from one sample.
Dynamic Response
Phenotyping, drug response, and other time-dependent biological measurements.
Distributed Deployment
High-capability systems outside centralized laboratory infrastructure.
Develop assays without building new instruments
Guanine applies the Nano-Bio-Info Scaling Law through a shared architecture that expands the amount, diversity, and temporal resolution of biological information extracted from a sample.
Instead of building a new instrument for each assay, OEMs can translate biomarkers directly into deployable assays on a common platform.
Shared platform
One architecture supports multiple assay classes and product configurations.
No new hardware per assay
Assay development is separated from instrument development.
Reusable deployment model
Systems can expand across applications, settings, and markets without redesign.
| Traditional Approach | Guanine Platform |
|---|---|
| Build instrument + assay together | Develop assay on existing platform |
| Hardware validation for each new system | Reuse validated platform architecture |
| No reuse across assays | Shared architecture reused across assays |
| Single-use system economics | Multi-assay platform economics |
| Capital-intensive deployment | Lower-cost scalable deployment |
A platform model replaces an instrument model.
The same architecture that expands capability also collapses cost
When hardware is no longer the limiting layer, development time, capital requirements, and deployment costs all change structurally.
This is not just a performance improvement. It is a new economic model for assay development and scale.
$6K–$8K class systems instead of $250K–$1M+ instrument stacks for advanced multi-omic measurement.
Assays deploy on a shared architecture rather than through new hardware cycles.
Lower-cost systems support broader placement across clinical and distributed settings.
Each new assay increases platform utilization rather than creating a new hardware silo.

A single platform across previously separate domains
The same system can support applications that existing infrastructure fragments into separate categories.
Low-abundance protein biomarkers
Detect and quantify low-concentration host-response signals and advanced protein panels.
Rare cells and low-concentration pathogens
Measure dilute targets from large sample volumes.
Genetic targets and resistance markers
Scale multiplex panels without increasing hardware complexity.
Multi-analyte measurement
Combine host, pathogen, protein, nucleic acid, and related signals from one sample.
Time-dependent biological response
Measure dynamic treatment response, cellular activity, and phenotypic change.

One architecture. Multiple configurations.
The platform can be configured for near-patient use, higher-density multiplex analysis, dynamic time-series measurement, and distributed deployment.
Point-of-Care
Lower-cost systems for decentralized and near-patient deployment.
Multiplex Analyzer
Higher-density configurations for advanced panels and broader assay menus.
Time-Series System
Dynamic measurement for response-based assays and functional biology.
All configurations share the same underlying architecture.

Standardize how advanced assays are built
Guanine does not just enable assays. It standardizes the path from biomarker to deployed product.
Assay translation
Convert biomarkers into deployable assay formats on a shared platform.
Workflow and cartridge integration
Unify preparation, enrichment, and measurement in a practical format.
Validation on established architecture
Reduce development uncertainty by building on an existing measurement system.
Each new assay compounds platform value
OEMs do not switch platforms for incremental improvements. They switch when one system replaces multiple constrained systems and enables assays that were not previously practical.
Each new assay increases system utilization, expands consumables revenue, and extends the relevance of the platform across adjacent markets.
This is not a better way to run existing assays. It is a way to build and scale assays that were not previously practical.


